Site Management Organization at Chiron Medical
Site Management Redefined for Clinical Excellence.
Chiron Medical CRO is committed to redefining the standards of site management to achieve clinical excellence. Our SMO unit specializes in providing tailored site management solutions that ensure every clinical site operates at the highest level of efficiency, compliance, and quality. From site selection and initiation to ongoing monitoring and patient recruitment, we manage every step with precision, allowing sponsors and investigators to focus on what matters most—delivering successful clinical outcomes.
Leveraging the extensive expertise of Chiron Medical CRO, our SMO offers an integrated approach that combines advanced technologies, regulatory expertise, and personalized site support. Our team of dedicated professionals works closely with each site to enhance performance, streamline operations, and maintain strict adherence to Good Clinical Practice (GCP) and local regulatory requirements.
"Site Management Redefined for Clinical Excellence" embodies our commitment to delivering superior site management services that drive clinical trial success. As your trusted partner, Chiron Medical CRO ensures that every site is managed with the utmost care, integrity, and expertise, setting a new standard in clinical research support.

Precision Site Management for Clinical Excellence.

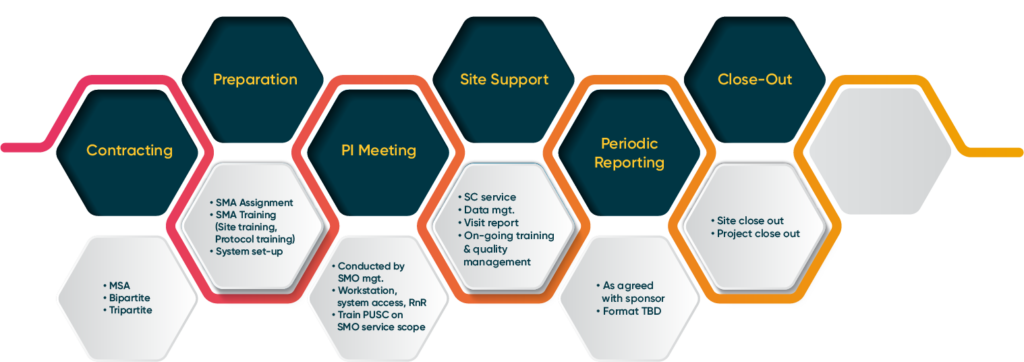
Our Expertise
SMO operates through three distinct units that together make up the cornerstones of our success, all driven by our commitment to advancing medical knowledge:
- Site Management Team: By guaranteeing smooth site operations, our skilled SMO staff maximizes all clinical trial-related aspects. We are prepared to expedite your study, offering everything from assistance with subject enrollment to comprehensive Clinical Research Coordinator services and accurate data entry.
- Site Start-Up Unit (SSU): We accelerates the clinical trial process through efficient site initiation, managing everything from feasibility assessments to regulatory submissions and site activation. With a proactive approach and strong relationships with regulatory authorities, the SSU ensures a smooth, compliant startup phase, achieving first dosing milestones within five weeks. Sponsors can rely on the SSU to start their trials on time while adhering to the highest standards of quality and compliance, ultimately driving clinical success.
- SMO Operations: Our SMO operations focus on delivering high-quality clinical trial support through a comprehensive suite of services. This includes rigorous site selection and feasibility assessments, streamlined site initiation and activation, and structured site management and monitoring. Our proactive approach to patient recruitment and retention ensures optimal enrollment, while robust quality assurance processes maintain compliance with Good Clinical Practice (GCP) and local regulations. By leveraging advanced technology for real-time data management and reporting, we provide sponsors with detailed insights into site performance, ultimately driving the successful execution of clinical studies and advancing medical research.
Our Impact
Our mission is to redefine the standards of site management in clinical research. Our impact is profound and multifaceted, driving significant advancements in the healthcare landscape:
Accelerating Clinical Trials: We streamline site operations to expedite patient recruitment and retention, ensuring that clinical trials are conducted efficiently and within timelines. Our focus on optimizing site performance contributes to faster delivery of new therapies to patients.
Strengthening Partnerships: By fostering strong collaborations with sponsors, clinical research organizations, and healthcare providers, we create a cohesive network that enhances the clinical trial process. Our collaborative approach ensures that all stakeholders are aligned towards common goals.
Empowering Local Sites: We invest in local sites by providing training, resources, and support. Our commitment to capacity building not only enhances the skill set of clinical staff but also contributes to the sustainability of research in local communities.
Upholding Quality Standards: Quality and compliance are at the core of our operations. We adhere to stringent regulatory requirements and industry best practices, ensuring the integrity of clinical data and the safety of study participants.
Innovating with Technology: Leveraging advanced technology and data analytics, we enhance operational efficiency, monitor trial progress in real time, and improve decision-making. Our tech-driven solutions support sites in managing resources effectively and maximizing trial success.
Creating Lasting Impact: Our dedication to ethical practices and patient-centered research ensures that our contributions extend beyond clinical trials. We strive to make a positive difference in the lives of patients and communities, ultimately leading to improved healthcare outcomes.
At CMSMO, we are not just managing sites; we are shaping the future of clinical research, committed to making a meaningful impact in the world of healthcare.
Our Services
Our SMO Division provides a wide range of services to help you maximize your clinical research experience. These include:
- The Scope of Services Provided By A
Clinical Research Coordinator: We make sure that every part of your study runs smoothly by providing careful schedule management, subject recruitment, AE/SAE reporting, and more. - Site Management Scope: Our site management scope covers everything from data entry to equipment management, ensuring the smooth functioning of your trials.
- Reporting Scope: We guarantee your study’s compliance by helping with all aspects of reporting, including protocol deviation reporting and EC and TMMDA initial/amendment submissions.
- Contact Scope: With a dedicated contact with SMO team, we provide 24/7 support for clinical trial stakeholders, offering real-time inquiry resolution, regulatory compliance assistance, and comprehensive services for site-related issues, including patient recruitment and protocol clarification. Our focus is on maintaining clear communication, ensuring privacy, and enhancing operational efficiency across all clinical activities.