Clinical Site Feasibility
With over 15 years of experience in conducting clinical trials locally, Chiron Medical has the know-how and means to carry out thorough protocol, site, and country feasibility studies that result in precise and reliable research designs.
Beyond departmental boundaries, we bridge the gap between your study and the ideal investigators. Leveraging our extensive network and proactive engagement, we navigate the intricacies of both internal and surgical departments within Medical Faculties and Training and Research Hospitals. This deep understanding translates into precise feasibility assessments, maximizing your chances of success on Turkish soil.
Our team’s in-depth knowledge of patient recruitment and retention tactics, regional regulatory requirements, clinical trial protocol design, and standards of care gives us unmatched insight into potential obstacles to successful clinical trial execution.
Chiron Medical uses a broad range of skills in addition to conventional clinical development planning to develop strategies for decentralized or hybrid clinical trials that are tailored to the best patient recruitment and retention tactics in order to reduce patient burden and maximize study engagement. Regardless of the sort of study you are conducting, we can provide you with the best clinical trial protocol, study design, and primary investigators using our proactive business intelligence tools and expert insights.

Feasibility Process
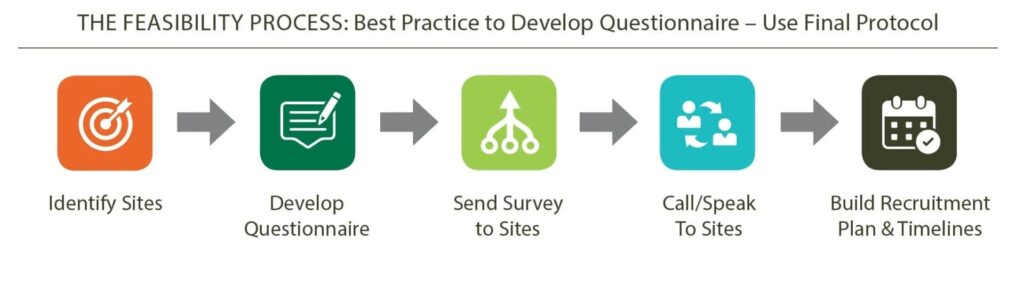

Strategic Feasibility, Seamless Trials.
Planning for Strategic Trials
Chiron Medical uses the following important data points to inform its planning and decision-making:
- Inclusion/exclusion criteria and endpoints
- Relationships with Key Opinion Leaders (KOLs) for all therapeutic areas
- The competitive environment and past enrollment rates
- Approval status
- Site ranking determined by regulatory findings and study experience
- Patient population at the site and community factors
- Investigator’s protocol-specific input
- Realistic study start-up timeline at each site
Available From Us
Using data from earlier research, Chiron Medical’s trial database, our therapeutic knowledge, and business intelligence technologies, we provide:
- A comprehensive overview of the local indication environment
- Suggestions for enhancing the design of the protocol, including an evaluation of the protocol’s impact in consultation with KOLs
- Medical issues and available options for the patient population
- The implementation of a thorough patient recruitment, diversity/inclusion, and retention plan that integrates patient voice, patient burden concerns, and patient journey mapping
- Expert comments on research administration and methodology, including suggestions for locations and deadlines, regulatory issues, and the best principal investigators to choose
- A thorough plan with risk assessment and mitigation strategies that will guarantee study delivery on schedule and within budget

Turning Challenges into Strategic Opportunities in Clinical Trials.

Strategic Feasibility, Turkish Expertise: Elevate Your Clinical Trials in Turkiye
Site Feasibility and Site Selection in Turkiye
Chiron Medical provides site selection and feasibility services for clinical studies conducted in Turkiye. Our services for feasibility consist of:
- Creation of site feasibility surveys for specific clinical studies with the guidance of potential experienced investigators.
- Search and identifying potential sites in Turkiye
- Distribution and gathering of completed surveys for feasibility
- Evaluations of the feasibility questionnaire and preliminary selection of Turkish sites
- Site qualification visits (SQVs) and reports, both on-site and remote
- Perform site feasibility and selection in Turkish cities with over 9,000 investigators in Chiron Medical database and potential clinical research sites/ hospitals spread over 81 cities with a combined population of about 86 million. The Turkish cities with the largest populations of potential patients are Istanbul (17 million), Ankara (6 million), and Izmir (5 million).
- Phase I-IV clinical trials with investigational medicinal product (IMP) including bioavailability and bioequivalence studies or medical devices in all therapeutic areas
- Providing services for biotech and pharmaceutical companies as well as academic sponsors
If you require assistance with the selection and feasibility of clinical sites in Turkiye, kindly get in touch with us. We will be pleased to provide you the top possible study sites, along with experienced investigators.
